Equipment Compatibility Assessment
A structured pre-commercial assessment for manufacturers evaluating whether existing plastics processing equipment can support a transition from conventional materials to biodegradable polymer compounds.
What this beta assessment is for
This beta version is designed to test the assessment workflow and report structure with early users before broader release.
Clarify whether the declared material transition appears technically feasible under the current processing configuration.
Identify likely thermal, flow, or mechanical constraints before committing to pilot trials or commercial-scale implementation.
Receive a PDF technical hypothesis report for internal decision support and early validation planning.
What the report helps clarify
The assessment is intended to give manufacturers a structured starting point for material transition discussions, not to replace material testing or specialist validation. Examples include flow instability, sealing difficulty, thickness variation, dimensional drift, heat resistance limitations, and surface quality changes.
GO / Conditional GO / HOLD
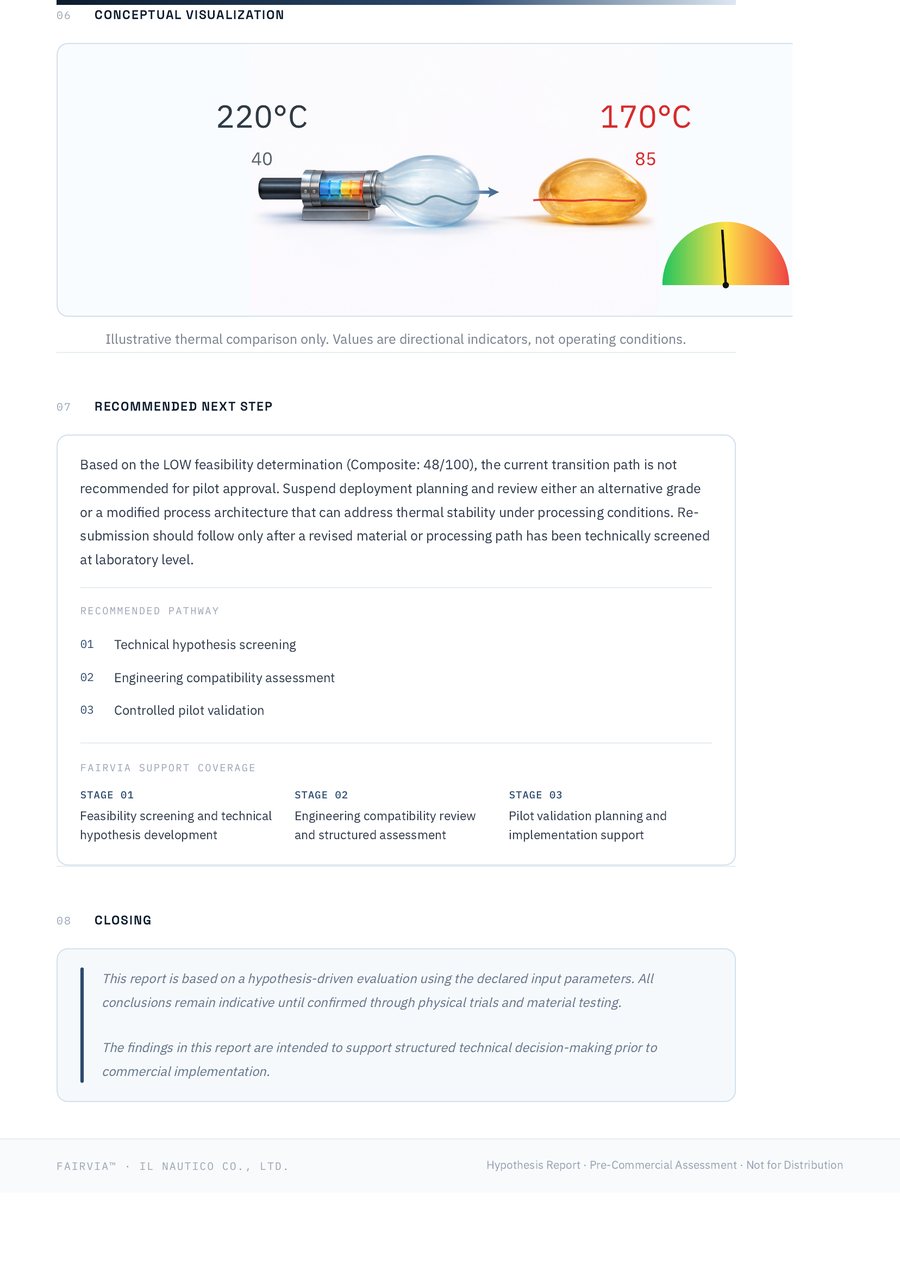
A feasibility band based on thermal, flow, and mechanical screening logic.
Main technical blocker
Identifies the most likely constraint affecting production stability or product realization.
Next validation step
Provides a practical direction for pilot validation, reassessment, or further technical review.
Actual PDF output examples
The beta assessment generates a PDF report structured around feasibility level, processing behaviour, failure analysis, quality assessment, and recommended next steps.

Designed for early-stage technical review
The beta form is designed to reduce unnecessary disclosure while still collecting the information required for a useful technical screening.
Company-identifying information is optional
Company name and contact person fields are optional. A valid email address is required only for report delivery and follow-up communication. You may submit the beta assessment without disclosing company-identifying information at this stage.
No sensitive formulation required
You are not required to submit confidential formulations, proprietary recipes, or detailed production know-how. The beta assessment is based on application, current material, target material, processing method, and major technical concerns.
Security, confidentiality, and purchase notes
Please review the following notes before proceeding with beta access.
Confidential handling
Submitted technical information is handled confidentially and used only for diagnostic report generation and related follow-up communication.
Technical hypothesis, not certification
The report provides a technical hypothesis and decision-support assessment. It does not constitute material certification, regulatory approval, production guarantee, or legal advice.
Payment and delivery
After payment, a secure assessment form link will be sent by email. After the form is submitted, the PDF report will be generated and delivered to the email address provided.
Beta participation
This beta access is offered to selected early users for validation of the assessment workflow and report structure. FairVia™ may request feedback after delivery.
Review the beta form before payment
The beta assessment form is shown below so that technical teams can confirm the required information before proceeding. Company-identifying information remains optional; a valid email address is required for report delivery.
Beta access for early technical evaluation
Use this beta access if you want to evaluate the assessment workflow before committing to a full Equipment Compatibility Assessment.
One-time payment. After payment, the secure form link will be sent by email. No company-identifying information or sensitive formulation details are required at this stage.